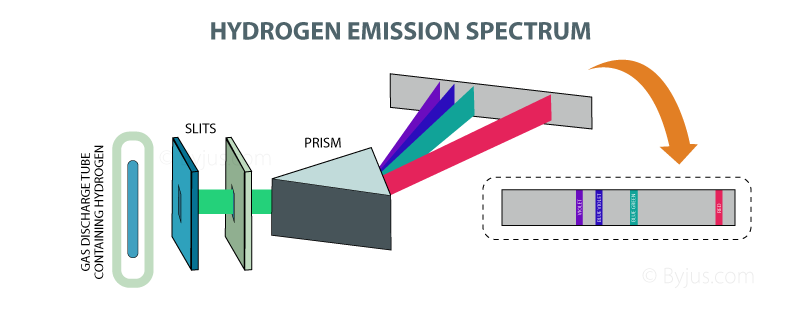
This means that line spectra can be used to identify elements.Ĭontinuous spectra are produced by electrons being shared between many atoms, giving a huge range of possible frequencies, as shown below. As a result each produces photons with different energy and so the line spectra for different elements will be different. These observed spectral lines are due to the electron making transitions between two energy levels in an atom. This is not a continuous spectrum as only light of specific frequencies and specific colours are produced.ĭifferent types of atoms have different energy levels. The emission spectrum of atomic hydrogen has been divided into a number of spectral series, with wavelengths given by the Rydberg formula. 1 2 3 4 5 6 Line emission and absorption spectra Studying the line spectra produced by hot gases and absorbed by cooler gases allows us to identify the elements in stars. This causes line emission spectra to be produced, as shown below. This means that each electron transition will produce a photon of a different frequency and hence a different colour. \(f\) is the frequency of light producedĪs the energy levels have different values, each of the possible electron transitions within an atom will produce a photon with a different energy.If an electron moves from level \(E_\) the energy of the photon can be worked out using the following: The energy of the photon can be worked out using the equation Hydrogen Line Spectrum The emission spectrum of atomic hydrogen is divided into a number of spectral series, with wavelengths given by the Rydberg formula. The amount of energy it loses will be equal to the difference in the energy levels it moves between. 1.3: Photoelectric Effect Explained with Quantum Hypothesis 1.5: The Rydberg Formula and the Hydrogen Atomic Spectrum Overview To introduce the concept of absorption and emission line spectra and describe the Balmer equation to describe the visible lines of atomic hydrogen. Below the text is a picture of an emission spectrum in the form of a black rectangular bar with five thin colored lines to represent emission lines.  
If an electron is in an excited state it can return to a lower energy level. An arrow points down from the gas cloud to text that reads, Emission Spectrum: Shows colored lines of light emitted by glowing gas.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
